Adverse Drug Reactions
Enquire nowAbout this book
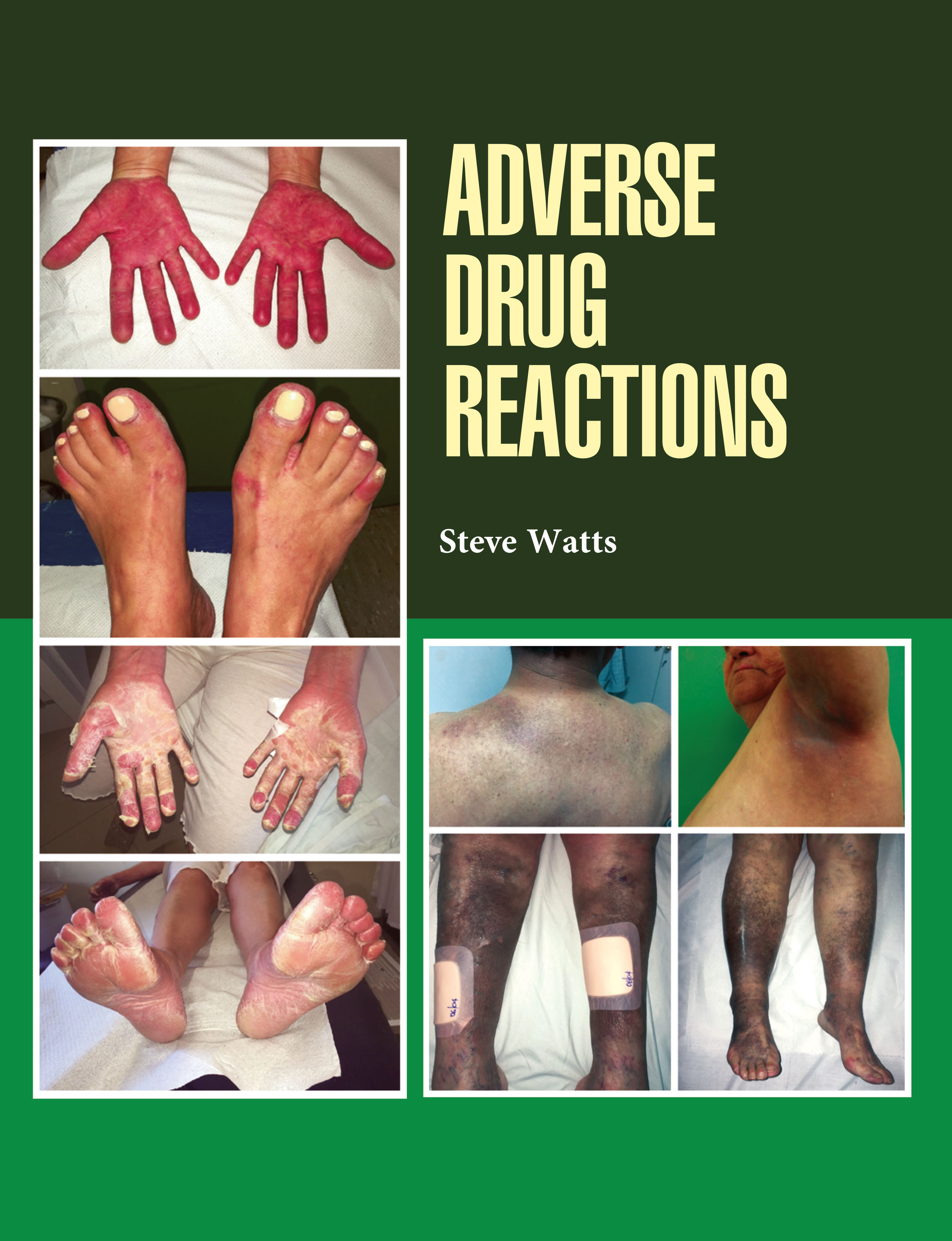
Adverse Drug Reactions (ADRs) are unintended, harmful, or unpleasant effects that occur after the administration of a drug at normal doses for therapeutic purposes. They represent a significant challenge in healthcare, as they can reduce treatment effectiveness, compromise patient safety, and increase healthcare costs. ADRs are classified into different types: Type A (augmented), which are predictable and dose-dependent, such as side effects or toxicity; and Type B (bizarre), which are unpredictable, idiosyncratic, or allergic reactions not related to the drug's known pharmacology. Other categories include Type C (chronic), Type D (delayed), and Type E (end-of-use or withdrawal reactions). The causes of ADRs are multifactorial, involving patient-related factors such as age, genetics, and underlying health conditions, as well as drug-related factors including dosage, interactions, and formulation. For example, elderly patients are more vulnerable due to altered drug metabolism and polypharmacy. Pharmacovigilance systems play a vital role in monitoring, detecting, and reporting ADRs to ensure drug safety. Clinicians must balance therapeutic benefits with potential risks by carefully prescribing and monitoring drugs. Ultimately, understanding ADRs is essential for improving patient care, guiding rational drug use, and supporting the development of safer medications through continuous research and regulatory oversight. Adverse Drug Reactions provides a comprehensive study of drug-related side effects, their mechanisms, classification, and impact on patient safety.







